
A jump through time – new technique rewinds the age of skin cells by 30 years
Key points:
- Research from the Babraham Institute has developed a new technique for rejuvenating skin cells. This technique has allowed researchers to rewind the cellular biological clock by around 30 years according to molecular measures, significantly longer than previous reprogramming methods.
- The partially rejuvenated cells showed signs of behaving more like youthful cells in experiments simulating a skin wound.
- This research, although in early stages, could eventually have implications for regenerative medicine, especially if it can be replicated in other cell types.
Research from the Babraham Institute has developed a method to ‘time jump’ human skin cells by 30 years, turning back the ageing clock for cells without losing their specialised function. Work by researchers in the Institute’s Epigenetics research programme has been able to partly restore the function of older cells, as well as rejuvenating the molecular measures of biological age. The research is published today in the journal eLife and whilst at an early stage of exploration, it could revolutionise regenerative medicine.
What is regenerative medicine?
As we age, our cells’ ability to function declines and the genome accumulates marks of ageing. Regenerative biology aims to repair or replace cells including old ones. One of the most important tools in regenerative biology is our ability to create ‘induced’ stem cells. The process is a result of several steps, each erasing some of the marks that make cells specialised. In theory, these stem cells have the potential to become any cell type, but scientists aren’t yet able to reliably recreate the conditions to re-differentiate stem cells into all cell types.
Turning back time
The new method, based on the Nobel Prize winning technique scientists use to make stem cells, overcomes the problem of entirely erasing cell identity by halting reprogramming part of the way through the process. This allowed researchers to find the precise balance between reprogramming cells, making them biologically younger, while still being able to regain their specialised cell function.
In 2007, Shinya Yamanaka was the first scientist to turn normal cells, which have a specific function, into stem cells which have the special ability to develop into any cell type. The full process of stem cell reprogramming takes around 50 days using four key molecules called the Yamanaka factors. The new method, called 'maturation phase transient reprogramming’, exposes cells to Yamanaka factors for just 13 days. At this point, age-related changes are removed and the cells have temporarily lost their identity. The partly reprogrammed cells were given time to grow under normal conditions, to observe whether their specific skin cell function returned. Genome analysis showed that cells had regained markers characteristic of skin cells (fibroblasts), and this was confirmed by observing collagen production in the reprogrammed cells.
Age isn’t just a number
To show that the cells had been rejuvenated, the researchers looked for changes in the hallmarks of ageing. As explained by Dr Diljeet Gill, a postdoc in Wolf Reik’s lab at the Institute who conducted the work as a PhD student: “Our understanding of ageing on a molecular level has progressed over the last decade, giving rise to techniques that allow researchers to measure age-related biological changes in human cells. We were able to apply this to our experiment to determine the extent of reprogramming our new method achieved.”
Researchers looked at multiple measures of cellular age. The first is the epigenetic clock, where chemical tags present throughout the genome indicate age. The second is the transcriptome, all the gene readouts produced by the cell. By these two measures, the reprogrammed cells matched the profile of cells that were 30 years younger compared to reference data sets.
The potential applications of this technique are dependent on the cells not only appearing younger, but functioning like young cells too. Fibroblasts produce collagen, a molecule found in bones, skin tendons and ligaments, helping provide structure to tissues and heal wounds. The rejuvenated fibroblasts produced more collagen proteins compared to control cells that did not undergo the reprogramming process. Fibroblasts also move into areas that need repairing. Researchers tested the partially rejuvenated cells by creating an artificial cut in a layer of cells in a dish. They found that their treated fibroblasts moved into the gap faster than older cells. This is a promising sign that one day this research could eventually be used to create cells that are better at healing wounds.
In the future, this research may also open up other therapeutic possibilities; the researchers observed that their method also had an effect on other genes linked to age-related diseases and symptoms. The APBA2 gene, associated with Alzheimer’s disease, and the MAF gene with a role in the development of cataracts, both showed changes towards youthful levels of transcription.
The mechanism behind the successful transient reprogramming is not yet fully understood, and is the next piece of the puzzle to explore. The researchers speculate that key areas of the genome involved in shaping cell identity might escape the reprogramming process.
Diljeet concluded: “Our results represent a big step forward in our understanding of cell reprogramming. We have proved that cells can be rejuvenated without losing their function and that rejuvenation looks to restore some function to old cells. The fact that we also saw a reverse of ageing indicators in genes associated with diseases is particularly promising for the future of this work.”
Professor Wolf Reik, who lead the research, said: “This work has very exciting implications. Eventually, we may be able to identify genes that rejuvenate without reprogramming, and specifically target those to reduce the effects of ageing. This approach holds promise for valuable discoveries that could open up an amazing therapeutic horizon.”
Notes to Editors
Publication reference
Gill et al. Multi-omic rejuvenation of human cells by maturation phase transient reprogramming, eLife, 2022
Press contact:
Honor Pollard, Communications Officer, honor.pollard@babraham.ac.uk
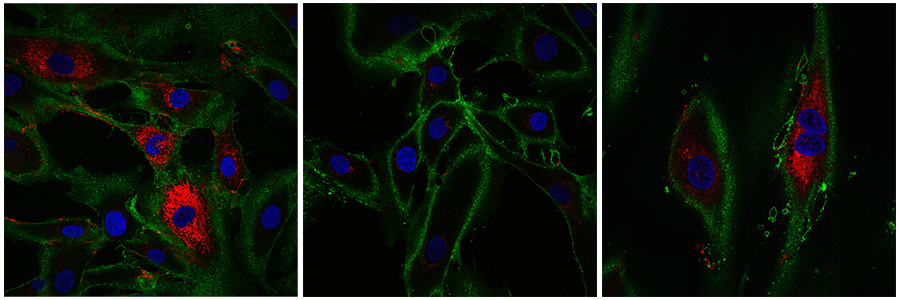
Image description: Left - fibroblasts from someone aged 20-22, middle - old fibroblasts that have not undergone reprogramming, right - reprogrammed cells. Collagen is shown in red
Affiliated authors (in author order):
Diljeet Gill – postdoctoral researcher, Reik lab,
Aled Parry - postdoctoral researcher, Reik lab
Fátima Santos - senior research scientist, Reik lab
Hanneke Okkenhaug, deputy manager, Imaging facility
Christopher Todd, postdoctoral researcher, Reik lab
Irene Hernando-Herraez - postdoctoral research scientist, Reik lab
Thomas Stubbs – Former PhD student in the Reik lab, now CEO, Chronomics Limited
Inês Milagre – Former Reik lab member, now postdoctoral fellow at the Laboratory for Epigenetic Mechanisms/Chromosome Dynamics Lab, Instituto Gulbenkian de Ciência
Wolf Reik – group leader, Epigenetics Programme. Director of Altos Labs Cambridge Institute since 1st March 2022
Research funding:
This work was funded by the Biotechnology and Biological Sciences Research Council, part of UK Research and Innovation (UKRI-BBSRC) and The Milky Way Research Foundation. Aled Parry is supported by a Sir Henry Wellcome Fellowship. Chris Todd is supported by the Wellcome Trust.
Additional/related resources:
Video animation about this work https://www.youtube.com/watch?v=NHK5w1sdr20
News, 14 August 2019: Pinpointing the molecular mechanisms of ageing
Royal Society Summer Science Exhibition Extra: Explore ageing with this interactive information on how the Institute’s cutting edge epigenetics research may be able improve the quality of life in old age. https://royalsociety.org/assets/sse-extra/index.html
About the Babraham Institute
The Babraham Institute undertakes world-class life sciences research to generate new knowledge of biological mechanisms underpinning ageing, development and the maintenance of health. Our research focuses on cellular signalling, gene regulation and the impact of epigenetic regulation at different stages of life. By determining how the body reacts to dietary and environmental stimuli and manages microbial and viral interactions, we aim to improve wellbeing and support healthier ageing. The Institute is strategically funded by the Biotechnology and Biological Sciences Research Council (BBSRC), part of UK Research and Innovation, through strategic programme grants and also receives funding from other UK research councils, charitable foundations, the EU and medical charities.
About BBSRC
The Biotechnology and Biological Sciences Research Council (BBSRC) is part of UK Research and Innovation, a non-departmental public body funded by a grant-in-aid from the UK government.
BBSRC invests in world-class bioscience research and training on behalf of the UK public. Our aim is to further scientific knowledge, to promote economic growth, wealth and job creation and to improve quality of life in the UK and beyond.
Funded by government, BBSRC invested £451 million in world-class bioscience in 2019-20. We support research and training in universities and strategically funded institutes. BBSRC research and the people we fund are helping society to meet major challenges, including food security, green energy and healthier, longer lives. Our investments underpin important UK economic sectors, such as farming, food, industrial biotechnology and pharmaceuticals.